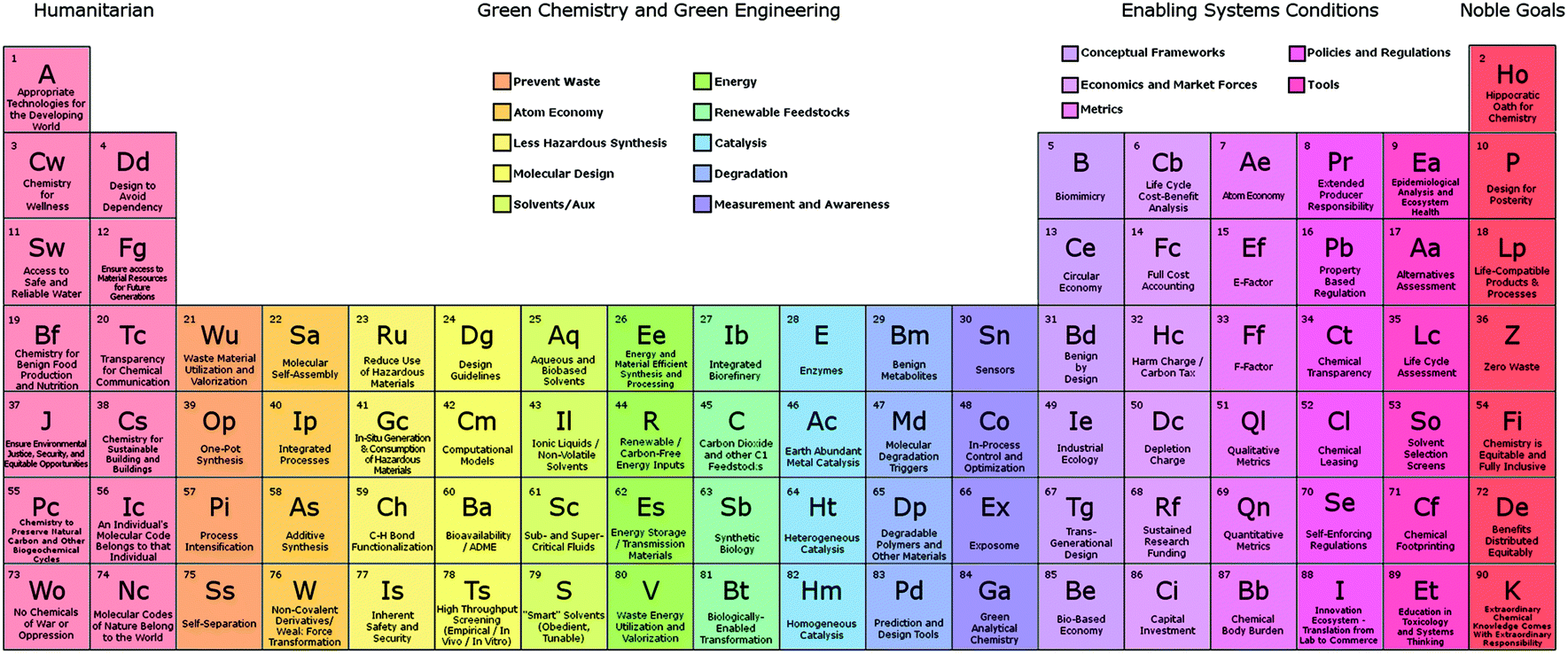
Yttrium aluminium garnet (YAG) laser, yttrium vanadate (YVO 4) as host for europium in television red phosphor, YBCO high-temperature superconductors, yttria-stabilized zirconia (YSZ) (used in tooth crowns as refractory material - in metal alloys used in jet engines, and coatings of engines and industrial gas turbines electroceramics - for measuring oxygen and pH of hot water solutions, i.e. Light aluminium-scandium alloys for aerospace components, additive in metal-halide lamps and mercury-vapor lamps, radioactive tracing agent in oil refineriesĪfter the village of Ytterby, Sweden, where the first rare-earth ore was discovered. Some of the rare-earth elements are named after the scientists who discovered them, or elucidated their elemental properties, and some after the geographical locations where discovered. This mineral was extracted from a mine in the village of Ytterby in Sweden four of the rare-earth elements bear names derived from this single location.Ī table listing the 17 rare-earth elements, their atomic number and symbol, the etymology of their names, and their main uses (see also Applications of lanthanides) is provided here. 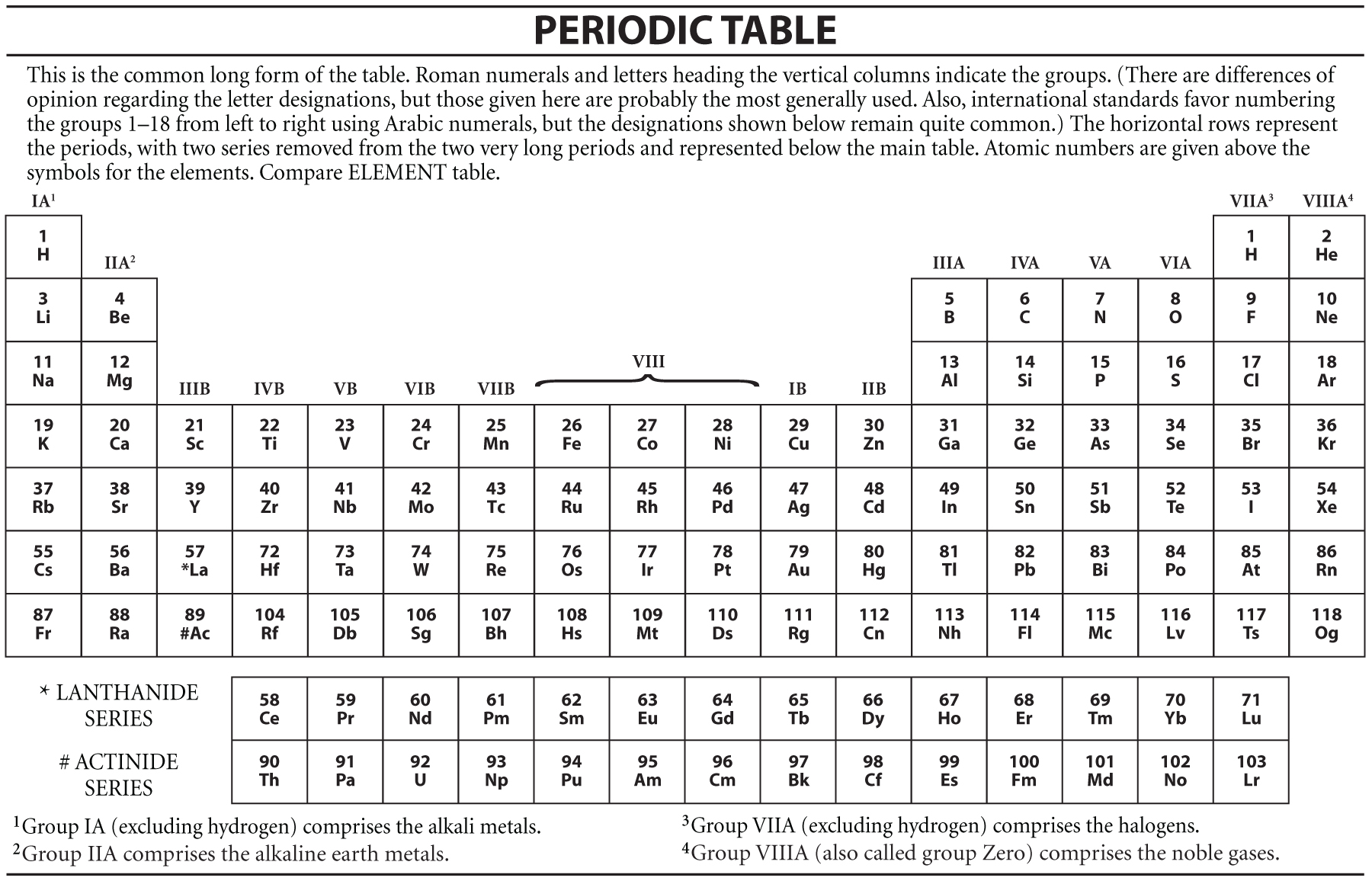
The first rare-earth mineral discovered (1787) was gadolinite, a black mineral composed of cerium, yttrium, iron, silicon, and other elements. Consequently, economically exploitable ore deposits are sparse. 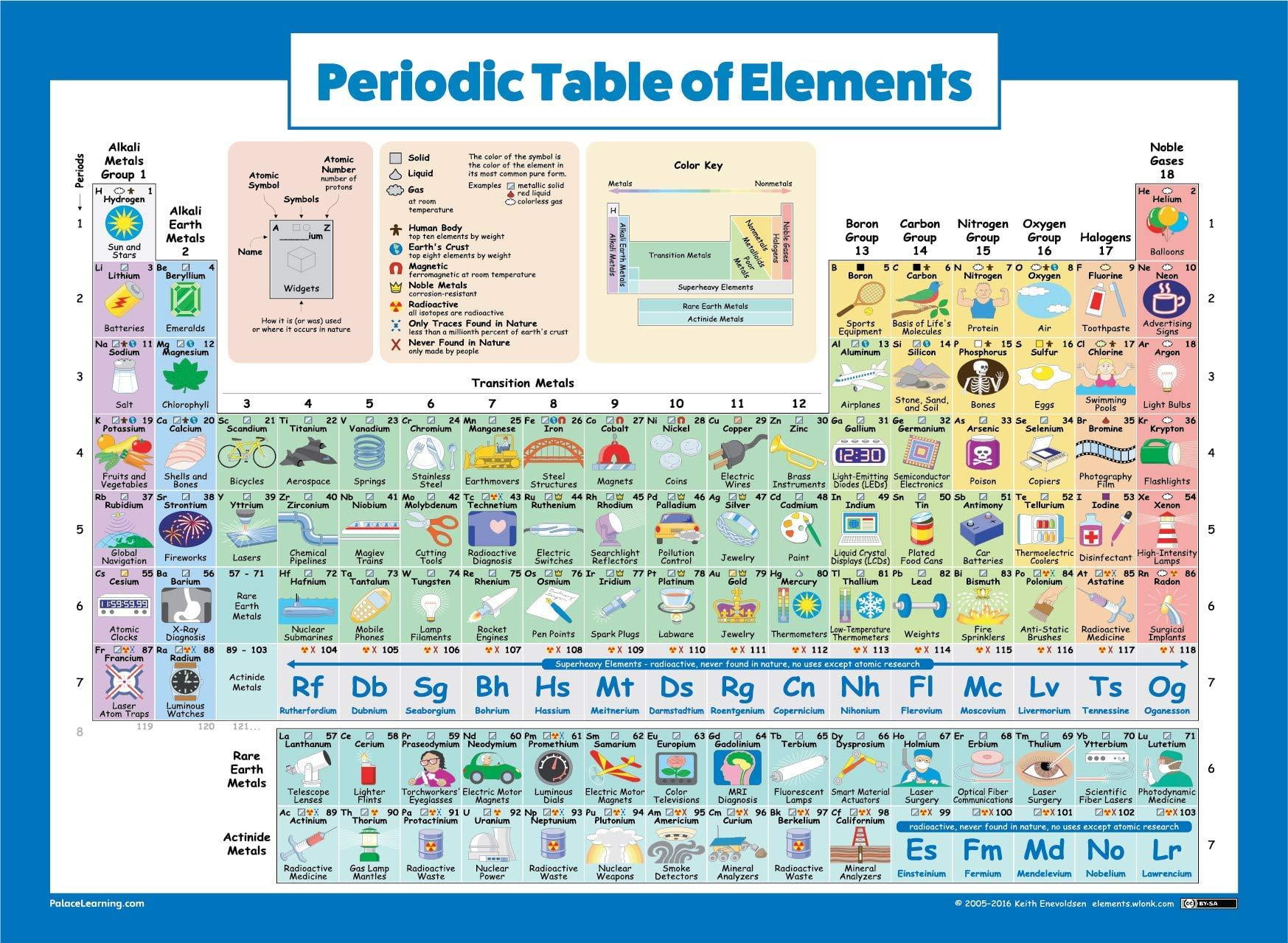
Though rare-earth elements are technically relatively plentiful in the entire Earth's crust ( cerium being the 25th-most-abundant element at 68 parts per million, more abundant than copper), in practice this is spread thin across trace impurities, so to obtain rare earths at usable purity requires processing enormous amounts of raw ore at great expense, thus the name "rare" earths.īecause of their geochemical properties, rare-earth elements are typically dispersed and not often found concentrated in rare-earth minerals. They are often found in minerals with thorium, and less commonly uranium. All isotopes of promethium are radioactive, and it does not occur naturally in the earth's crust, except for a trace amount generated by spontaneous fission of uranium-238. The water-soluble compounds are mildly to moderately toxic, but the insoluble ones are not. These elements and their compounds have no biological function other than in several specialized enzymes, such as in lanthanide-dependent methanol dehydrogenases in bacteria. They react with steam to form oxides and ignite spontaneously at a temperature of 400 ☌ (752 ☏). These metals tarnish slowly in air at room temperature and react slowly with cold water to form hydroxides, liberating hydrogen. The term 'rare-earth' is a misnomer because they are not actually scarce, although historically it took a long time to isolate these elements. Scandium and yttrium are considered rare-earth elements because they tend to occur in the same ore deposits as the lanthanides and exhibit similar chemical properties, but have different electrical and magnetic properties. Compounds containing rare earths have diverse applications in electrical and electronic components, lasers, glass, magnetic materials, and industrial processes. The rare-earth elements ( REE), also called the rare-earth metals or rare earths or, in context, rare-earth oxides, and sometimes the lanthanides (although yttrium and scandium, which do not belong to this series, are usually included as rare earths), are a set of 17 nearly indistinguishable lustrous silvery-white soft heavy metals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
